 The lessons learned in this post will continue through the study of chemical reactions and equations. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021. Visualize trends, 3D orbitals, isotopes, and mix compounds. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) 28.02 amu. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. concentration, fraction, ratio (molar, mass and. Rows are arranged so that elements with similar properties fall into the same vertical columns ('groups'). Interactive periodic table showing names, electrons, and oxidation states. The concept of molar mass can also be applied to compounds. The periodic table is a chart that organizes the elements by increasing atomic number and their chemical and physical properties (see article History of the Periodic table of elements). We also learned how to calculate the molar mass of a compound using the periodic table and how to convert mass into moles. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. We learned about Avogadro’s number and how it relates to the mole, which is a unit used to express the amount of a substance. In conclusion, understanding molar mass is an essential concept in chemistry as it allows us to relate the amount of a substance to the number of particles present in it. See original paper for the range of these elements from different sources Isotope-abundance variations and atomic weights of selected elements: 2016 (IUPAC Technical Report), Pure Appl. Specifically, the number is defined as 6.022 \times 10^. List of Elements with Range of Atomic Weights. Using the periodic table to find the mass for each mole of our elements we have: 1moleC 1 moleC ×(12.011gC 1 moleC) 12.011gC (8.3.8) (8.3.8) 1 m o l e C 1 m o l e C × ( 12.011 g C 1 m o l e C) 12.011 g C. 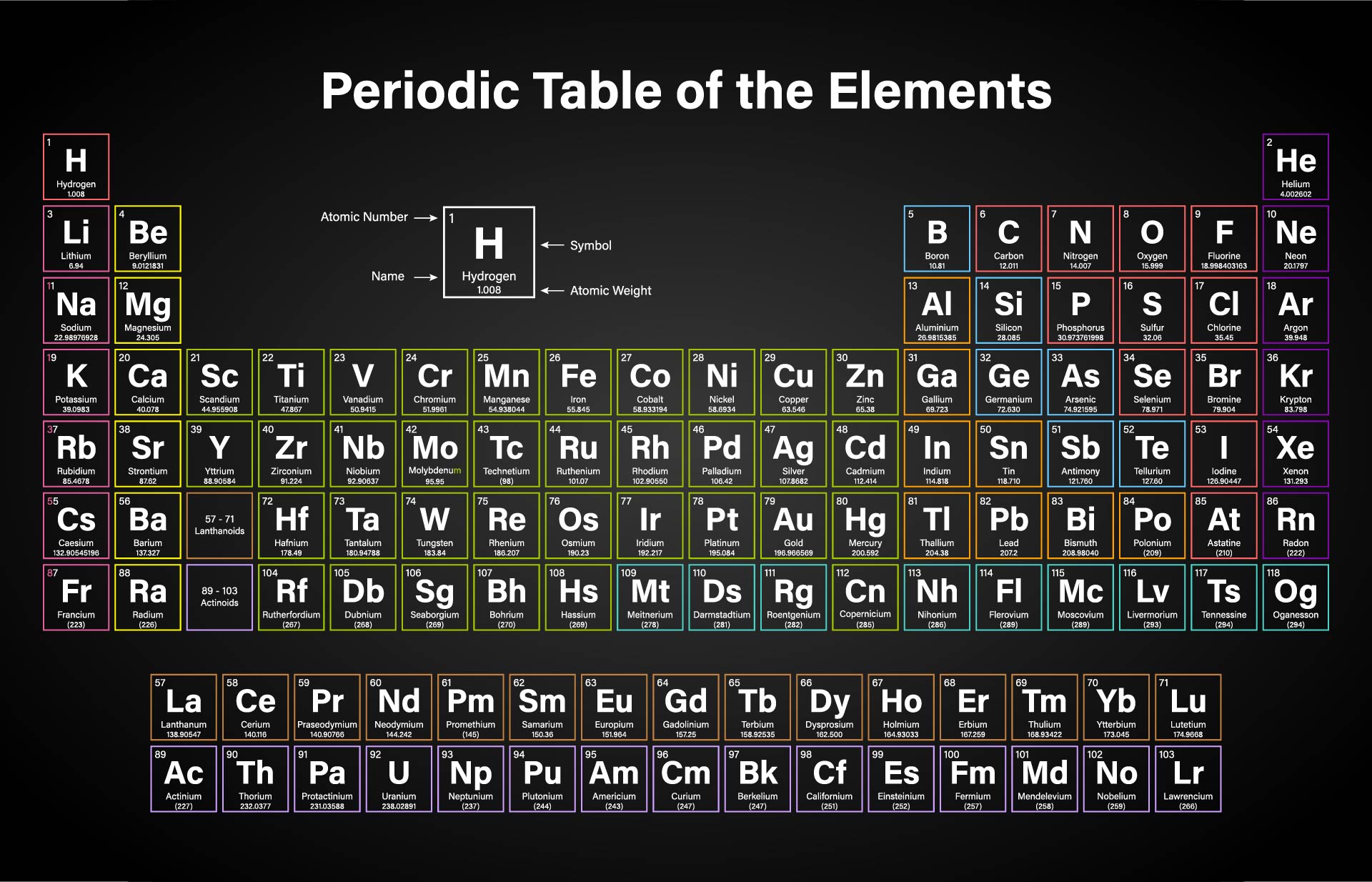
Example 3: Finding the Mass of a Number of MoleculesĪvogadro’s number is a fundamental constant that represents the number of particles (atoms, molecules, ions) in one mole of a substance. First we need to determine the mass of one mole of methane (CH 3 OH).How to Find Molar Mass Using the Periodic Table Find it by adding up the element atomic masses.The Mole and its Relationship to Avogadro’s Number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
